











| Model NO. | HS-230 | Application | Commercial Use |
|---|---|---|---|
| Portable | Portable Design | Certification | CE0197, ISO13485, FDA 510K |
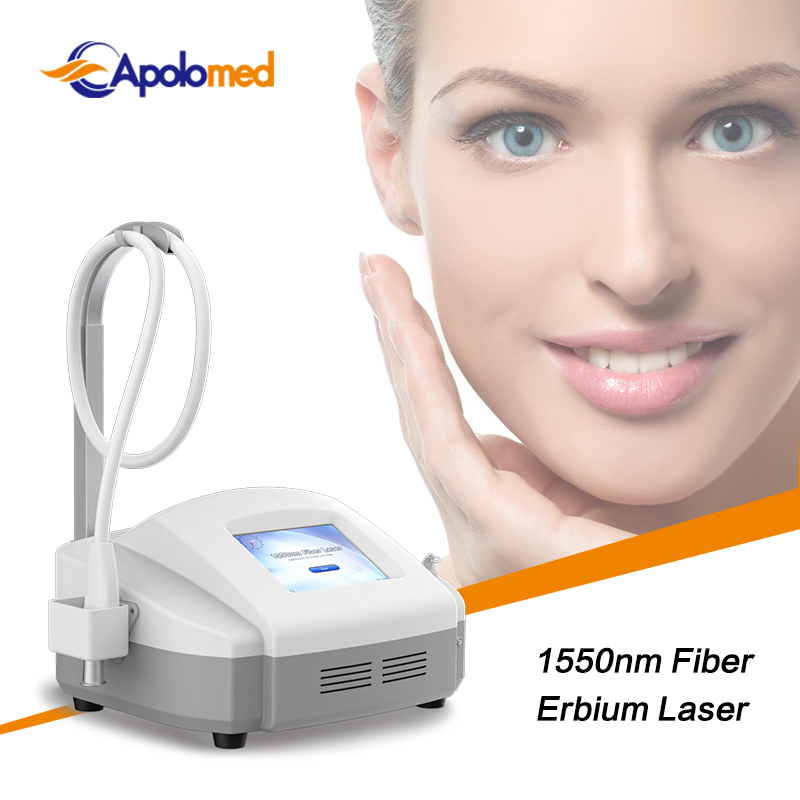
| Wavelength | 1550nm | Laser Power | 15W |
| Laser Output | 1~120mj/DOT | Scan Area | 20*20mm |
| Density | 25~3025 PPA/Cm2 | Pulse Width | 1~20ms/DOT |
| Cooling System | Advanced Air Cooling | Origin | China |
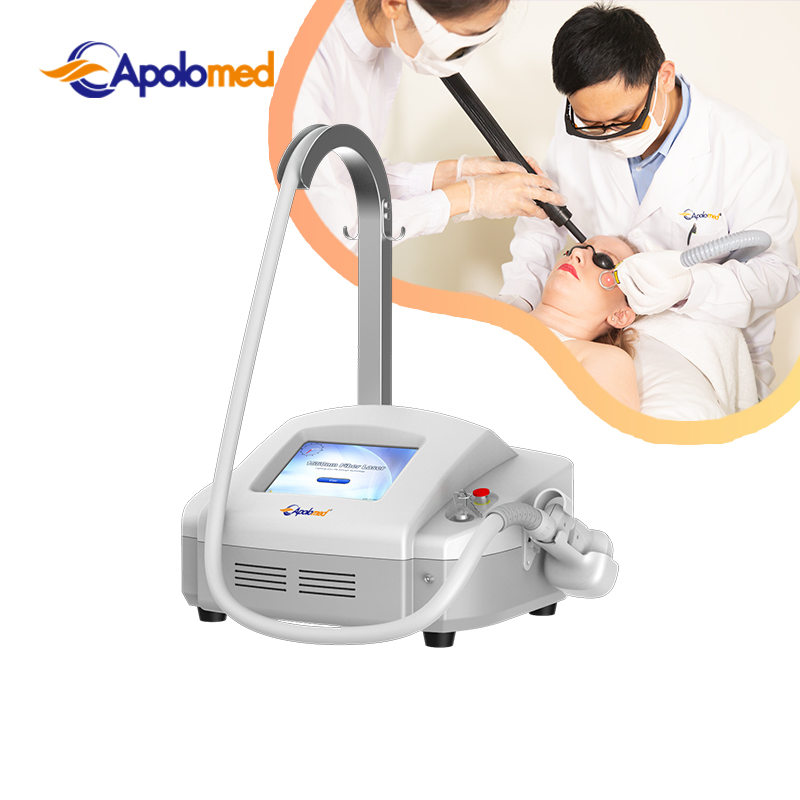
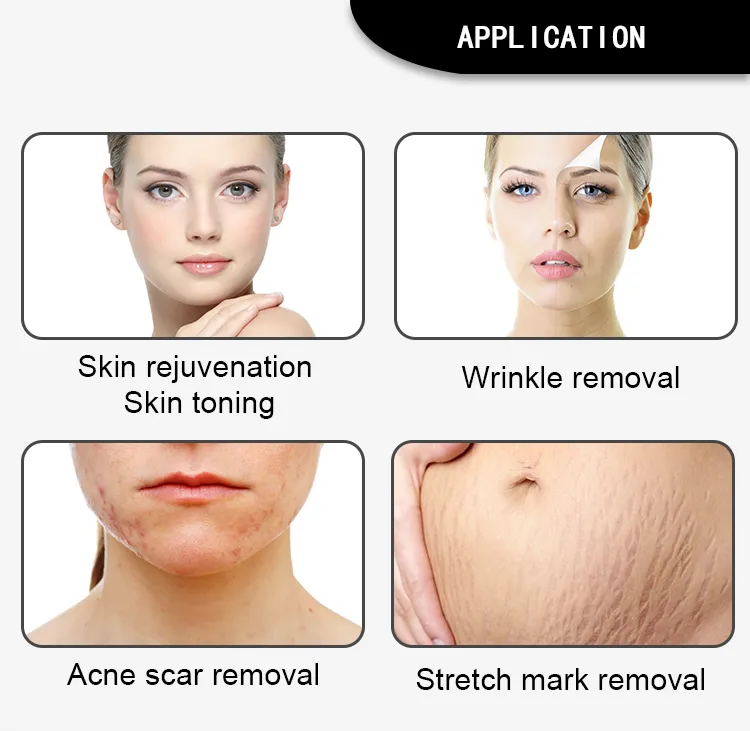
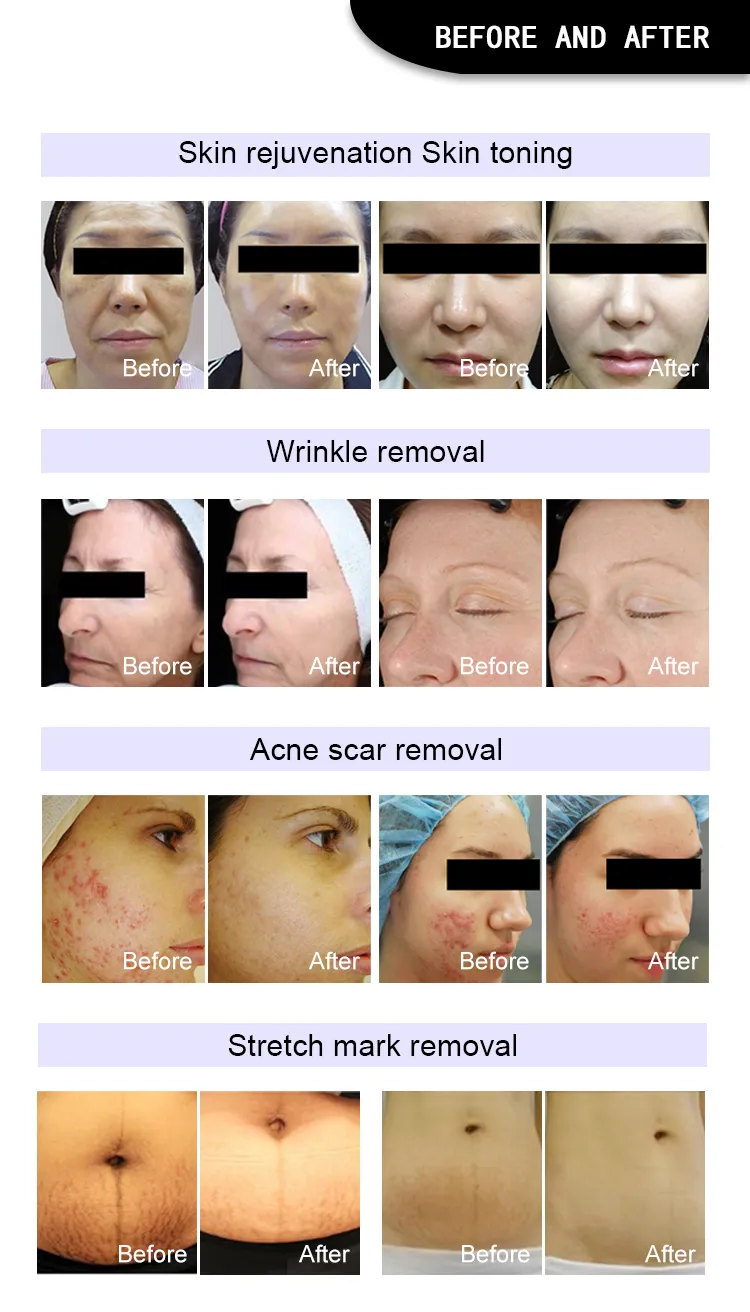
No Bleeding Erbium 1550 Laser Equipment Erbium Fiber Fractional Laser for Stretch Marks Removal
💡
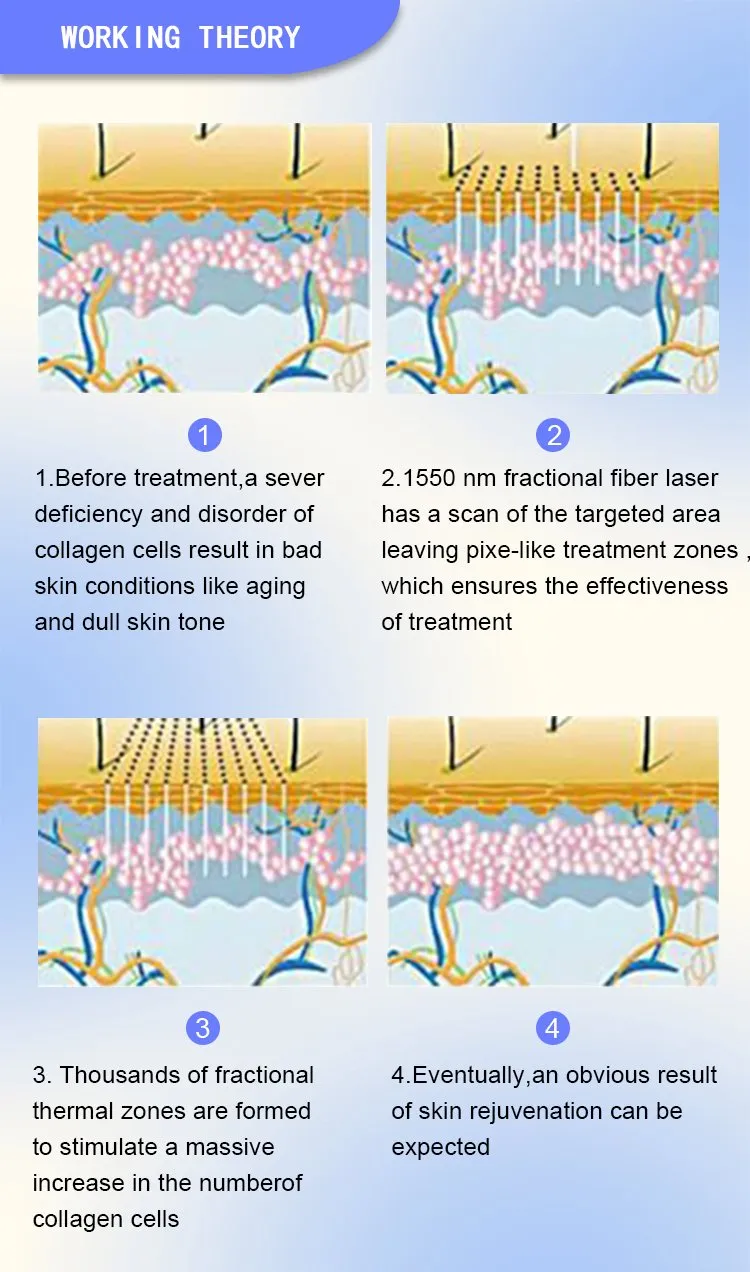
Principle of 1550nm Skin Rejuvenation Fiber Laser:
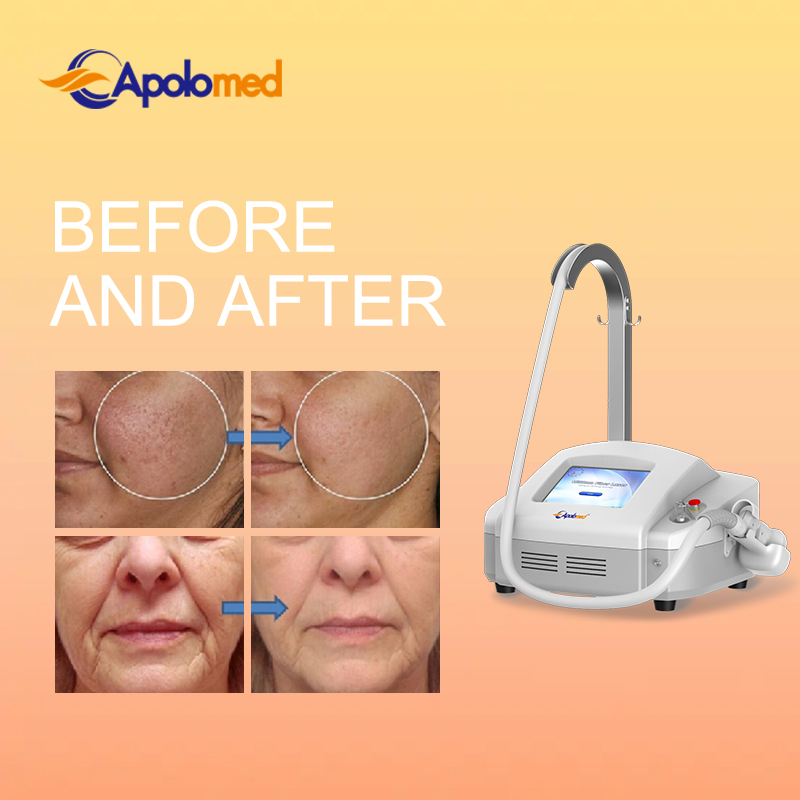
The 1550nm Erbium fiber laser is split into microscopic beams, creating microscopic thermal zones. This triggers the skin's natural healing process while protecting surrounding tissues. The treatment remolds collagen and stimulates skin regeneration by penetrating deep into the dermis. This fractional approach allows for faster healing, minimal downtime, and superior clinical results.

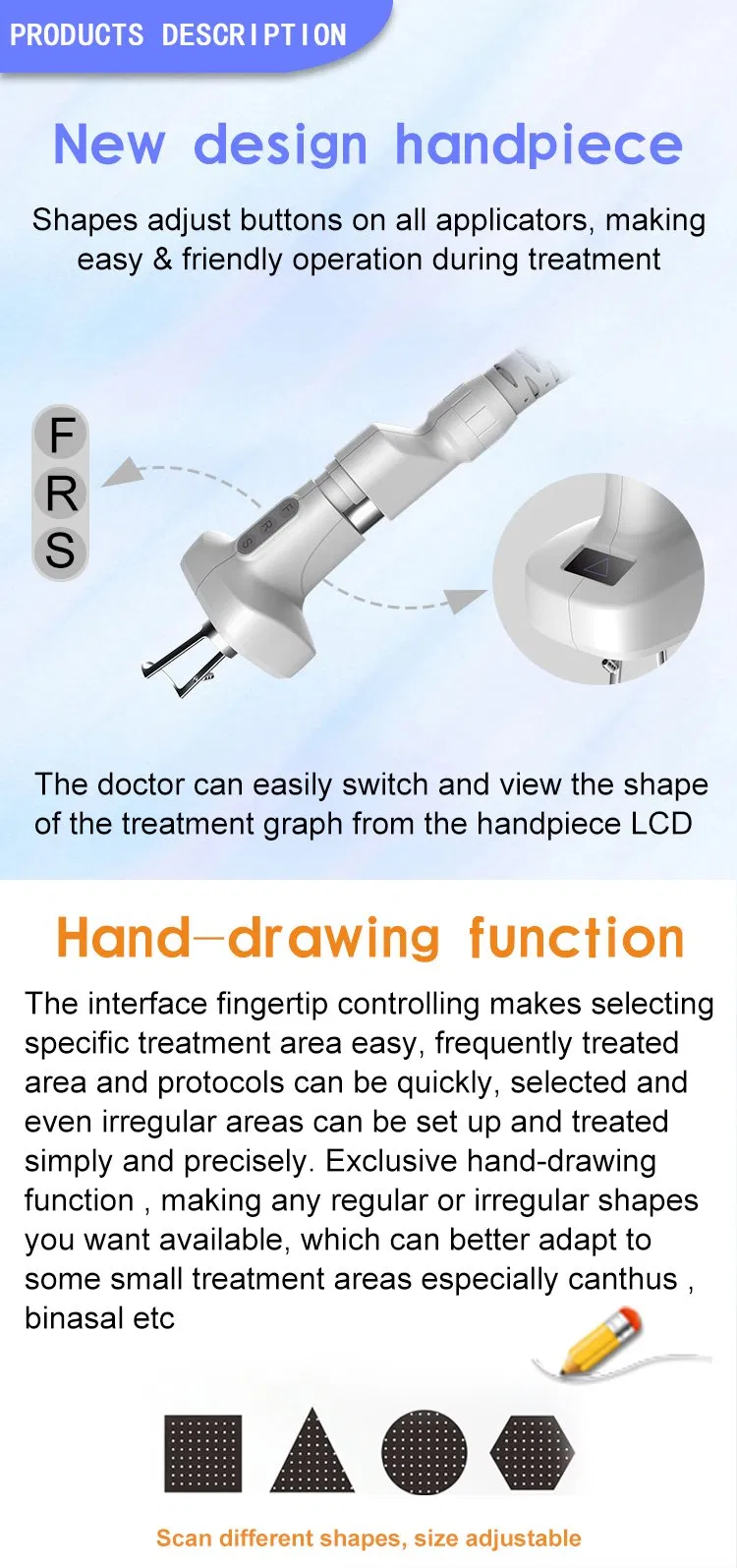



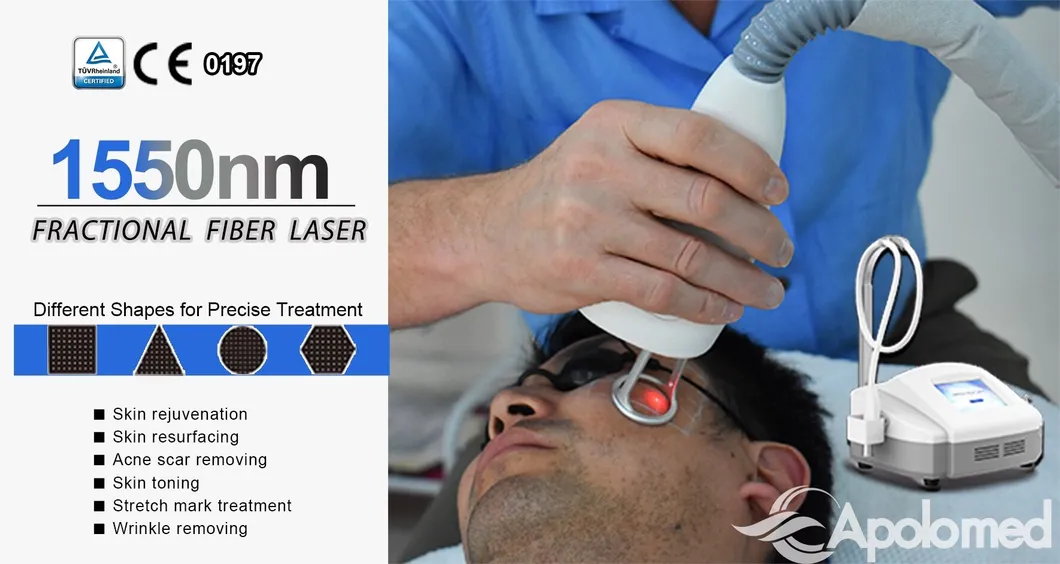
Free set scan shapes for precise treatment
| Model | HS-230 |
|---|---|
| Wavelength | 1550nm Erbium fiber laser |
| Laser Power | 15W |
| Laser Output | 1~120 mJ/dot |
| Density | 25~3025 PPA/cm2 (12 levels) |
| Scan Area | 20x20mm |
| Pulse Width | 0.06~8ms/dot |
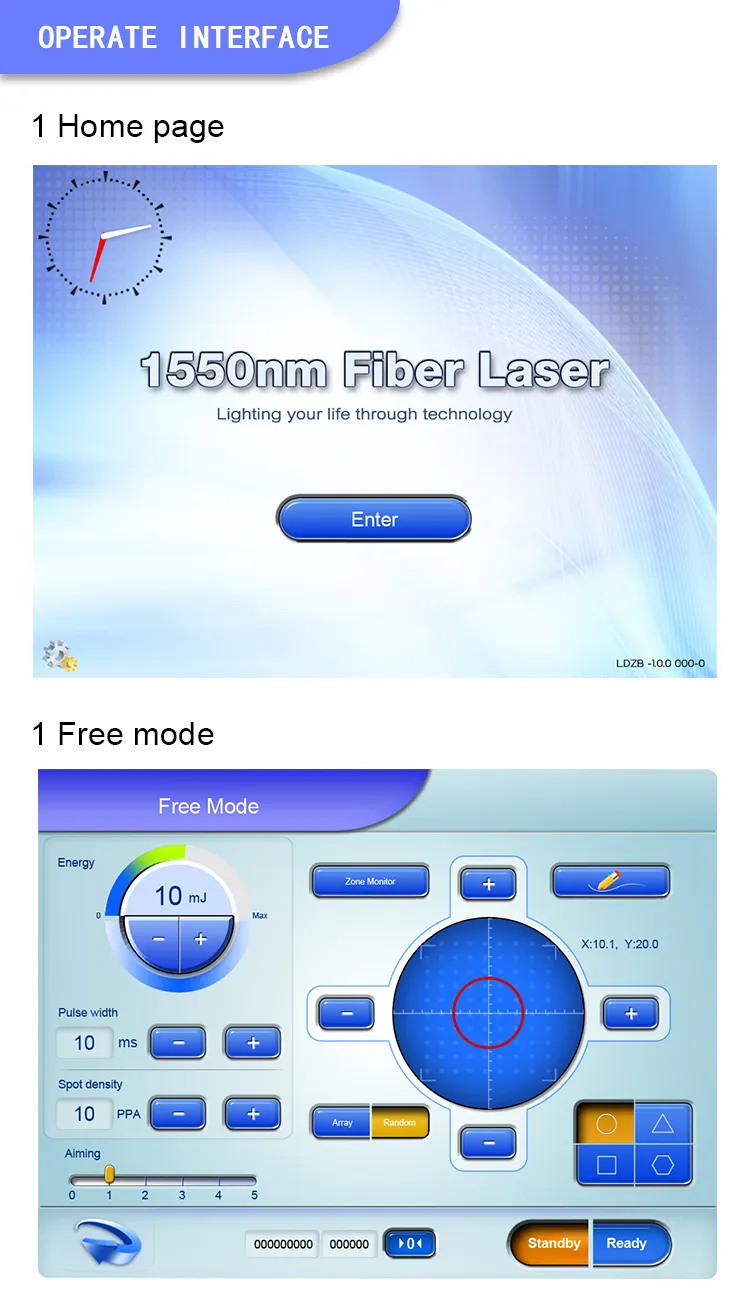
| Operate Interface | 9.7" True Color Touch Screen |
| Dimension | 52x44x32cm (L*W*H) |
| Weight | 30Kg |
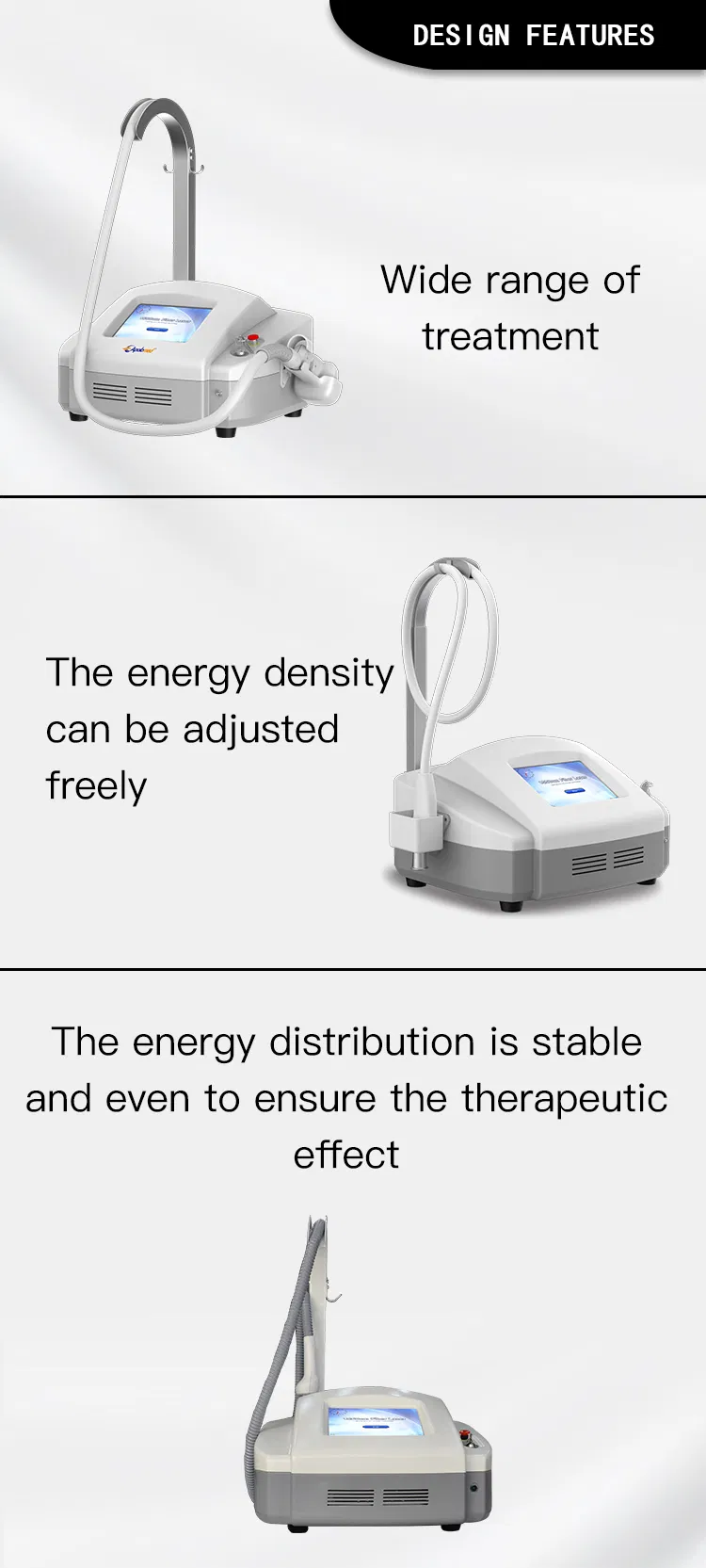
1 Wide range of indications with one compact machine.
2 Customizable treatment areas (including irregular shapes).
3 Fully adjustable energy density for tailored results.
4 Stable energy output ensuring clinical safety.
5 Android-based OS with customer database support.
6 RF ID/IC card management for versatile business models.
As a leading designer and manufacturer of aesthetic devices since 2001, our technologies include Pico Nd:YAG, CO2, HIFU, and PDT LED systems. We focus on high standards, with over 40 patented in-house devices designed to meet global beauty requirements.

Manufacturing strictly follows ISO 13485. Our products comply with Medical CE under MDD and MDR regulations. We have also obtained certificates such as US 510K, Australia TGA, and Brazil Anvisa.



