









| Parameter | Value |
|---|---|
| Model NO. | HS-890 |
| Application | Commercial Aesthetic Treatment |
| Portable Design | Yes |
| Certification | CE0197, ISO13485, FDA 510K |
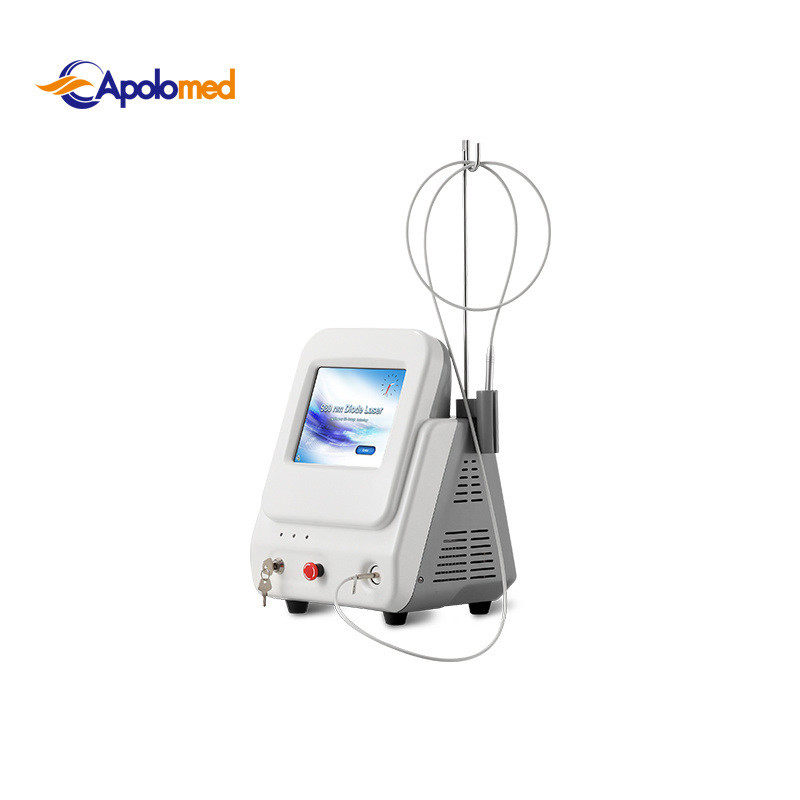
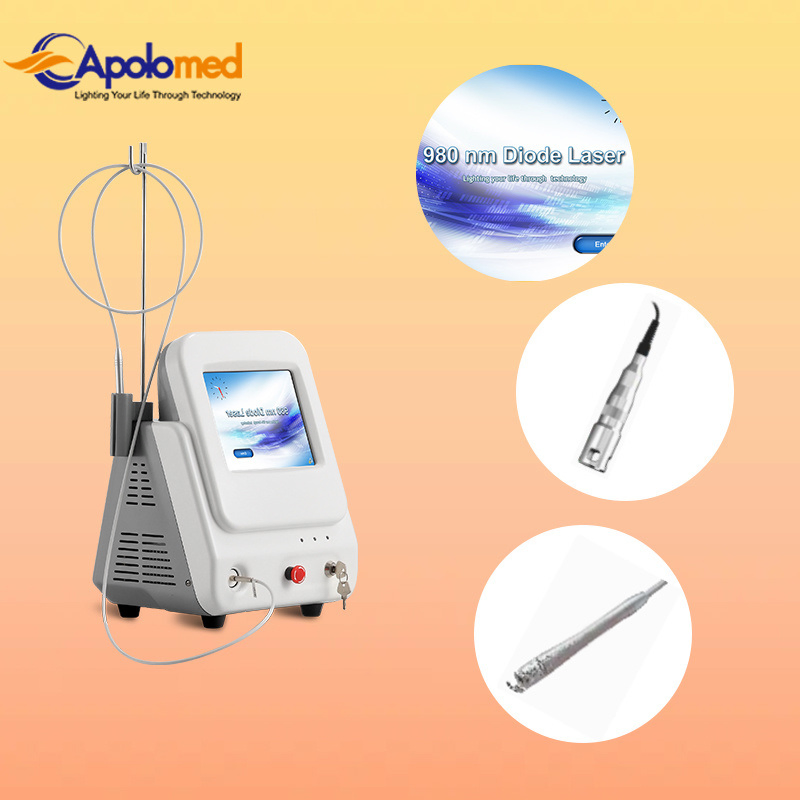
| Wavelength | 980nm |
| Output Power | 2~30W |
| Output Modes | CW, Single or Repeat Pulse |
| Pulse Width | 5~400ms |
| Cooling System | Air Cooling |
| Control Mode | 8" True Color Touch Screen |
| Power Supply | AC 100~240V, 50/60Hz |
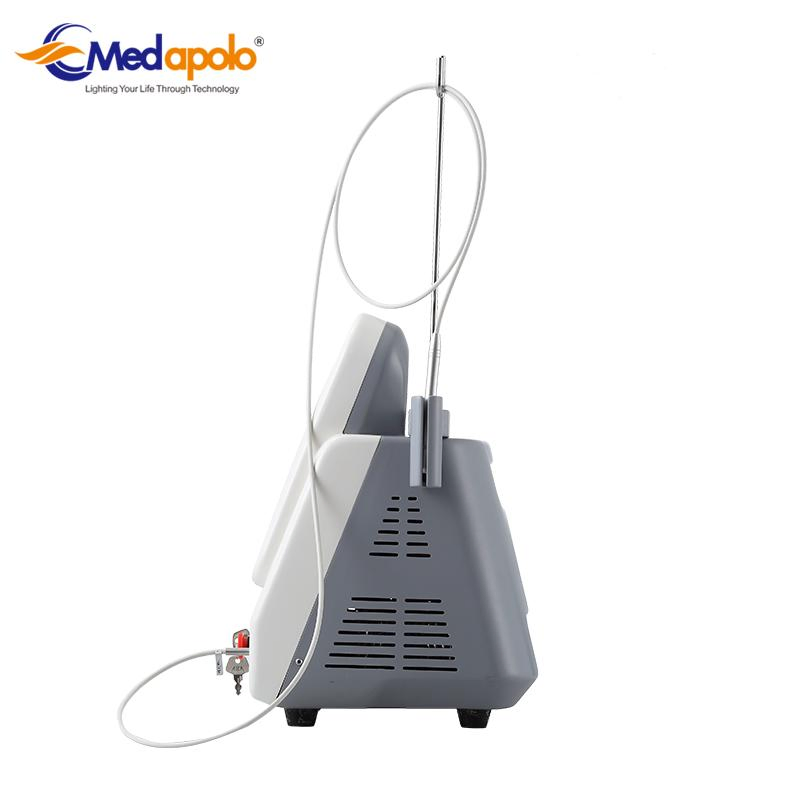
| Dimensions | 28*27*37cm (L*W*H) |
| Net Weight | 8Kgs |
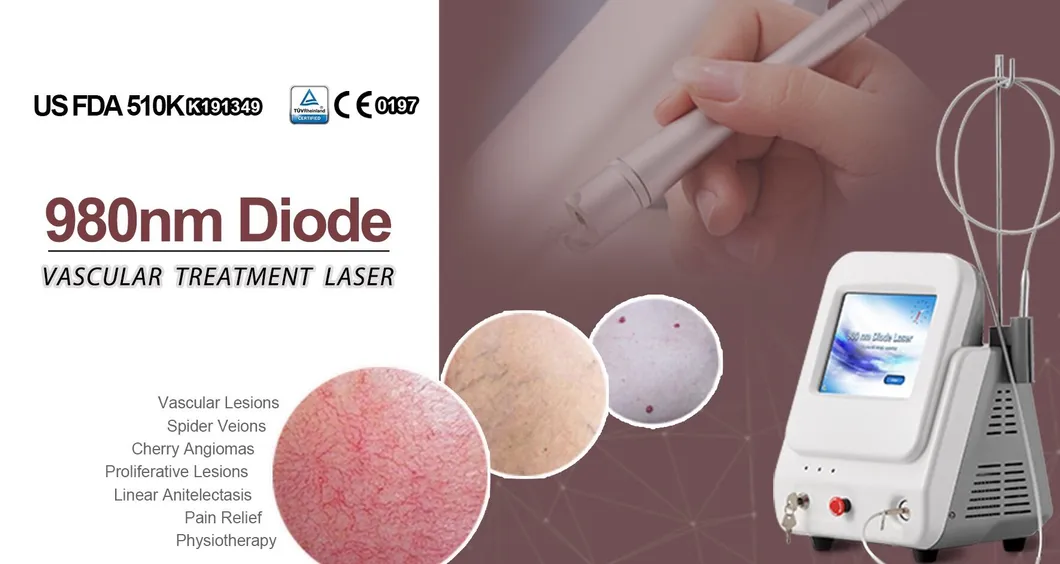
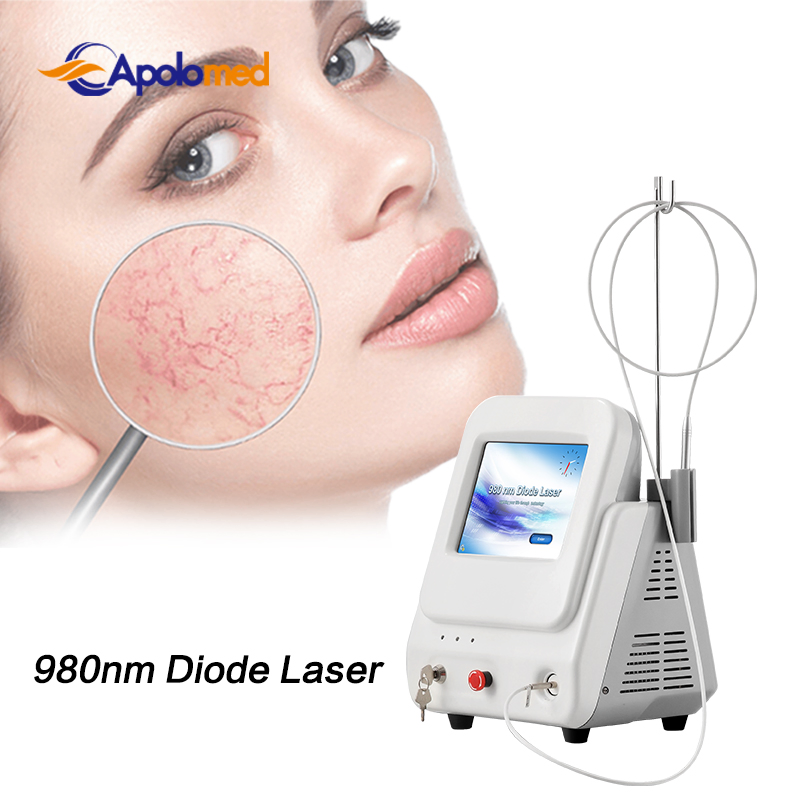
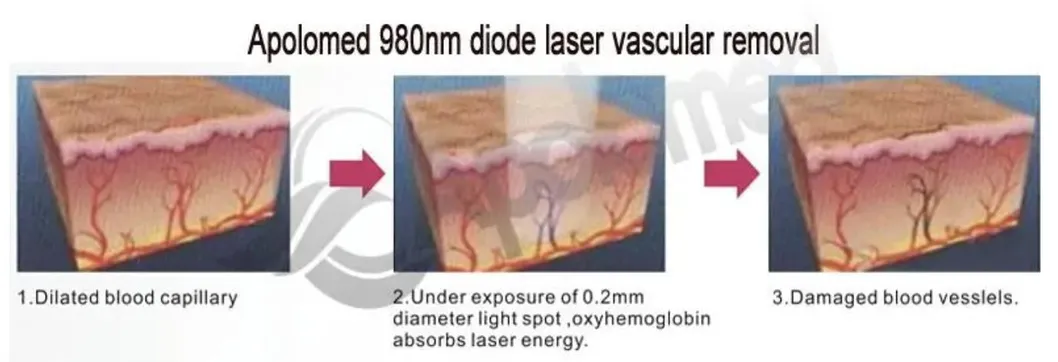
Based on the theory of "selective laser photothermal", the 980nm diode laser system utilizes a specific wavelength to penetrate the skin for targeted vascular treatment. Under laser irradiation, hemoglobin and red pigment in capillaries maximize absorption of the laser energy. This leads to solidification, blocking of blood vessels, and capillary shrinkage, resulting in intravascular coagulation and eventual metabolic degradation.
The specific 980nm wavelength ensures that the normal intact skin tissue architecture is preserved to the greatest degree during treatment, ensuring a high therapeutic effect without damaging the superficial skin layer.

| Laser Wavelength | 980nm |
| Laser Output Power | 2~30W |
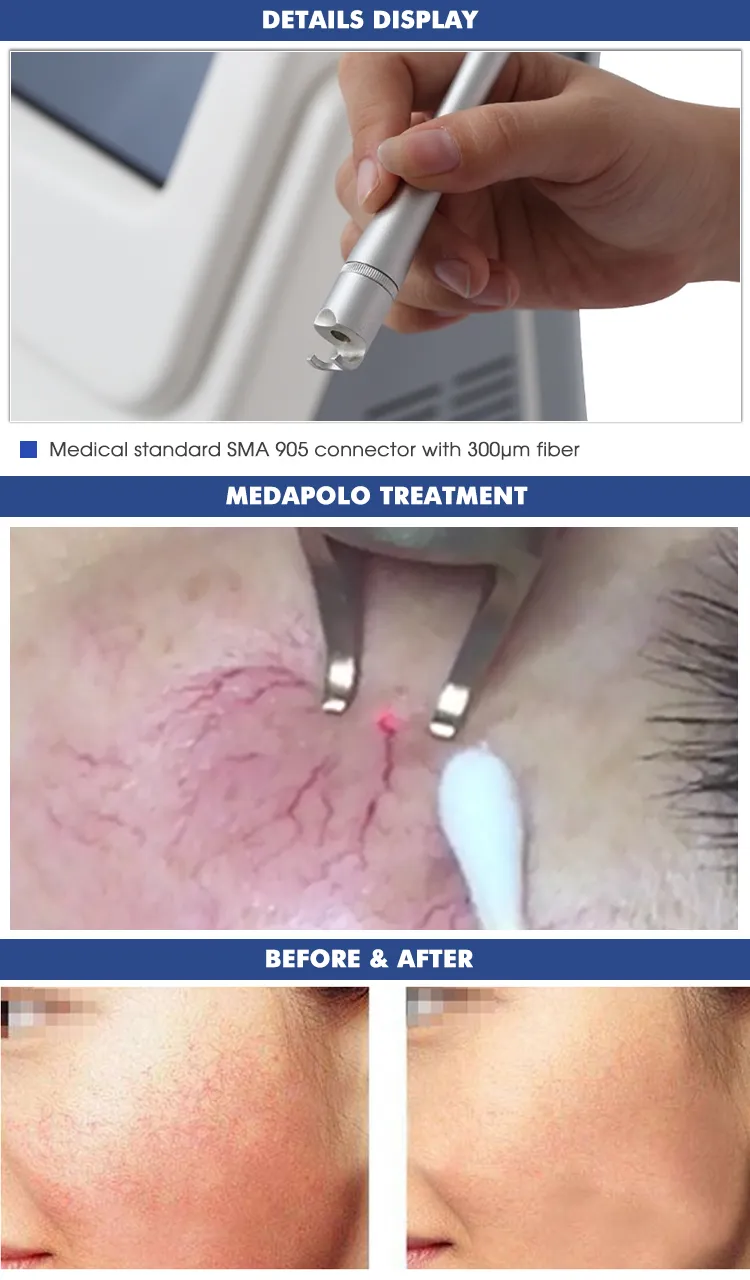
| Transmission System | Fibers of 300um, with SMAS 905 connector |
| Aiming Beam | Diode 650nm (red), ≤2mW |
| Pulse Repetition Rate | 1, 2, 3, 5, 10~50Hz |
| Single Pulse Energy | 0.1~12J |
| Repeat Pulse Power | 0.1~18W |
Equipment is manufactured strictly in accordance with ISO 13485 standards. Products comply with Medical CE certificates under Council Directive 93/42/EEC (MDD) and regulations (EU) 2017/745 (MDR). High-end devices have also obtained certificates from US 510K, Australia TGA, and Brazil Anvisa.


